1/8/2024 Shelby Lawson
Written by Shelby Lawson
Neurodegenerative disorders such as Alzheimer’s disease affect more than 270 million people worldwide. AD is the leading cause of dementia, resulting in memory loss due to atrophy of neurons in the hippocampus, which is the part of the brain that regulates learning and memory. Nanoparticles designed to carry drugs have emerged as a strategy for treating different diseases, but in the context of neurodegenerative disease, much of the research has focused on developing strategies for getting nanoparticles across the blood brain barrier and into targeted regions of the brain.

In a new study, an interdisciplinary team of researchers at the University of Illinois Urbana-Champaign have developed nanoparticles that are able to selectively bind to activated astrocytes and microglia cells that mediate brain inflammation in AD, and found that both AD and aging strongly affect the ability of nanoparticles to cross the BBB and localize to the hippocampus.
The BBB consists of a network of blood vessels surrounding the brain that tightly regulate what molecules (including drugs) can enter the brain. The BBB makes it difficult for nanoparticles carrying drugs to enter the brain, although nanoparticles can prevent the drugs from being “washed away”, or losing their activity along the way when passing through the BBB. However, research has suggested that the BBB weakens with AD and age. This inspired a team of researchers led by Joon Kong (M-CELS leader/EIRH/RBTE), a professor of chemical and biomolecular engineering, and Hee Jung Chung (M-CELS), an associate professor of molecular and integrative physiology, to synthesize a nanoparticle that could take advantage of this compromised BBB, and bind specifically to reactive astrocytes and microglia cells in the hippocampus of AD affected individuals.
“I think people have overlooked how the vascular permeability of the BBB changes with AD pathology,” said Kong. “We thought, instead of putting peptides or proteins onto nanoparticles that can help them penetrate the BBB, as others have done, let’s just make the nanoparticles small enough that they can take advantage of the leaky BBB, and engineer these particles in a way that lets them remain in the brain in a stable manner.”
The nanoparticles are designed to bind to CD44, a cell surface protein that is produced by reactive astrocytes and microglia cells, more than neurons, especially during neuroinflammation, a hallmark of AD-afflicted brain regions such as the hippocampus. The advantage of nanoparticles binding to these CD44-expressing cells is that the nanoparticles are retained longer in the hippocampus, rather than quickly being washed out, according to Kong.
The researchers injected the CD44 seeking nanoparticles into both older and younger mice that either had AD or were healthy. They then looked at the distribution of nanoparticles in the hippocampus across the treatments.
In the hippocampi of AD mice, they found high concentrations of nanoparticles regardless of age, though older AD mice had stronger concentrations than younger AD mice. The researchers say this was predicted, and further demonstrates that the BBBs of those with AD are considerably weakened. Not only were the nanoparticles able to penetrate the BBB, but they were also retained for longer in the hippocampus, for at least 2 hours post-injection, with preliminary data suggesting even longer retention.
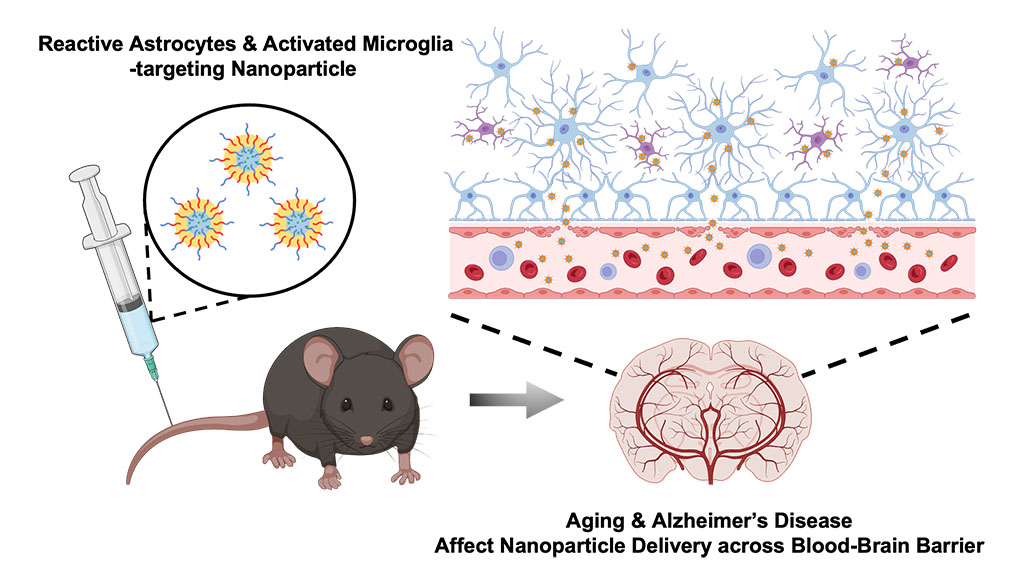 In the brains of healthy young mice, no nanoparticles were found, meaning that their BBBs were intact. However, to the team’s surprise, they found a significant amount of nanoparticles in the brains of healthy older mice, suggesting that the BBB weakens considerably with increasing age, even in those without AD.
In the brains of healthy young mice, no nanoparticles were found, meaning that their BBBs were intact. However, to the team’s surprise, they found a significant amount of nanoparticles in the brains of healthy older mice, suggesting that the BBB weakens considerably with increasing age, even in those without AD.
“This finding was surprising, because the older mice in this study are equivalent to a human age of only about 60 years old,” said Chung. “We knew there would be some leakiness of the BBB with age, but we thought there would be much less penetration of nanoparticles into the brain than we found. This highlights that there is age-dependent and disease-dependent penetration of the nanoparticles across the BBB to deep brain regions affected by AD.”
‘’This study offers valuable insights into advancing our understanding of nanoparticle transport to the brain in aging and Alzheimer's patients,” said Kai-Yu Huang, a graduate student in Kong’s lab. “It prompts us to think about the future strategies for the development of nano-scale drug carriers to target inflamed brain cells across different phases of aging-related brain disorders.’’
The next step is to try adding candidate drugs to the nanoparticles and see whether they could improve cognition and memory in AD mouse models, according to the researchers. They are also planning to measure how long their nanoparticles can be retained in the brain, which could help provide longer and more consistent drug delivery to patients treated with nanoparticles in the future. The team hopes this finding will provide a guideline for how to design drug carriers in the future for treating diseases, both within the brain and beyond.
“This extends beyond just the brain, because this technology can be used for other diseases, not just brain disease,” said Chung. “By modifying the surface moiety of nanoparticles, we can directly target different organs, given we know something specific to target within those organs. The use of nanoparticles in medicine has broad and innovative applications.”
This research was supported by the Alzheimer’s Disease Association, National Institutes of Health, the National Institute of Neurological Disorders and Stroke, the Chan Zuckerberg Biohub Chicago Acceleration Research Award, and the University of Illinois. The study can be found at https://doi.org/10.1021/acs.nanolett.3c03222